Description of Candesartan & Hydrochorothiazide
Candesartan Cilexetil & Hydrochorothiazide are angiotensin il type I (AT) receptor antagonist and a diuretic respectively used for the treatment of hypertension. Their fixed dose combination works by making your blood vessel relax & widen, thus helping to lower your blood pressure. The fixed dose combination has an additive effect on their respective antihypertensive properties.
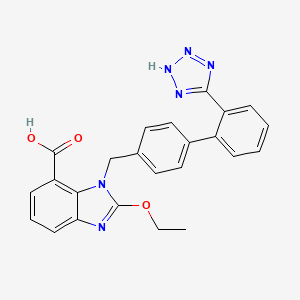
Candesartan cilexetil, a nonpeptide, is chemically described as (±)-1-Hydroxyethyl 2-ethoxy-1-[p-(O-1H-tetrazol-5-ylphenylbenzyl-7-benzimidazolecarboxylate, cyclohexyl carbonate (ester).
Fixed dose combination of Candesartan & Hydrochorothiazide is indicated for the treatment of essential hypertension in adults. It also indicated for treatment of adults with heart failure and impaired heart muscle function. In addition, it is used as add-on therapy to ACE inhibitor or when ACE inhibitors are not tolerated
How Candesartan & Hydrochorothiazide Work
Angiotensin II is formed from angiotensin I in a reaction catalyzed by angiotensin converting enzyme (ACE, kininase 11). Angiotensin II is the principal pressor agent of the renin-angiotensin system, with effects that include vasoconstriction, stimulation of synthesis and release of aldosterone, cardiac stimulation and renal reabsorption of sodium. Candesartan blocks the vasoconstrictor and aldosterone-secreting effects of angiotensin Il by selectively blocking the binding of angiotensin II to the AT, receptor in many tissues, such as vascular smooth muscle and the adrenal gland. Its action is, therefore, independent of the pathways for angiotensin Il synthesis.
Blockade of the renin-angiotesin system with ACE inhibitors, inhibits the biosynthesis of angiotensin II from angiotensin 1, which is widely used in the treatment of hypertension. ACE inhibitors also inhibit the degradation of bradykinin, a reaction also catalyzed by ACE, because candesartan does not inhibit ACE (kininase ), it does not affect the response to bradyknin. Whether this difference has clinical relevance is not yet known.
Candesartan does not bind to or block other hormone receptors or ion channels known to be important in cardiovascular regulation. Blockade of the angiotensin II receptor I I inhibits the negative regulatory feedback of angiotensin Il on renin secretion but the resulting increased plasma renin activity and angiotensin II circulating levels do not overcome the effect of candesartan on blood pressure.
Hydrochlorothiazide is a thiazide diuretic. Thiazides affect the renal tubular mechanisms of electrolyte reabsorption, directly increasing excretion of sodium and chloride in approximately equivalent amounts: Indirectly, the diuretic action of hydrochlorothiazide reduces plasma volume, with consequent increases in plasma renin activity, aldosterone secretion, urinary potassium loss and decreases in serum potassium levels, The renin-aldosterone link is mediated by angiotensin II, so co-administration of an angiotensin Il receptor antagonist tends to reverse the potassium loss associated with these diuretics. The mechanisms of the antihypertensive effect of thiazides is unknown.
Dosage And Administration of Candesartan & Hydrochorothiazide Combination
Dosage Forms & Strengths
candesartan/hydrochlorothiazide tablet
- 16mg/12.5mg
- 32mg/12.5mg
- 32mg/25mg
The usual recommended starting dose of candesartan cilexetil is 16 mg once daily, when it is used as monotherapy in patients who are not volume depleted. Candesartan cilexetil can be administered once or twice daily with total daily doses ranging from 8 mg to 32 mg. Patients requiring further reduction in blood pressure should be titrated to 32 mg. Doses larger than 32 mg do not appear to have a greater blood pressure lowering effect.
Hydrochlorothiazide is effective in doses of 12.5 to 50 mg once daily.
The recommended daily dose of Candesartan & Hydrochorothiazide fixed dose combination is one tablet once daily. Antihypertensive effect is usually attained within 4-5 weeks of initiation of treatment.
To minimize dose-independent side effects, it is usually appropriate to begin combination therapy only after a patient has failed to achieve the desired effect with monotherapy. Candesartan & Hydrochorothiazide combination should be taken once daily with or without food. Its efficacy and safety in children has not been established
Clinical Pharmacology of Candesartan & Hydrochorothiazide
Pharmacokinetics
Candesartan cilexetil
Candesarten cilexetil is rapidly and completely bioactivated by ester hydrolysis during absorption from the gastrointestinal tract to candesartan a selective AT1 subtype -angiotensin II receptor antagonist.
Candesaran is mainly excreted unchanged in urine and feces (via bile). It undergoes minor hepatic metabolism by O-deethylation to an inactive metabolite. The elimination half-life of candesartan is approximately 9 hours. After single and repeated administration, the pharmacokinetics of candesartan is linear for oral doses up to 32 mg. Candesartan and its inactive metabolite do not accumulate in serum upon repeated once-daily dosing. Following administration of candesartan, cilexetil, the absolute bioavailability of candesartan was estimated to be 15%. After tablet ingestion, the peak serum concentration (C) is reached after 3 to 4 hours Food with a high fat content does not affect the bioavailability of candesartan. The apparent volume of distribution is 0.8 kg.
Hydrochlorothiazide
Hydrochlorothiazide is rapidly absorbed from GI tract with a bioavailability of approximately 70% The plasma protein binding of hydrochlorothiazide is approximately 50% The apparent volume of distribution is 0.8 l/kg. Hydrochlorothiazide is not metabolized and it is excreted entirely unchanged. The plasma half-life has been observed to vary between 56 and 14.8 hours. After administration of hydrochlorothiazide with candesartan cilexetil, no additional accumulation of hydrochlorothiazide occurs after repeated doses of combination compared to monotherapy.
Metabolism and Excretion
Candesartan cilexetil
Total plasma clearance of candesartan is 0.37 ml/min/kg with a renal clearance of 0.19 ml/min/kg. When candesartan is administered orally, about 26% of the dose is excreted unchanged in urine and bile and to minor extent eliminated by hepatic metabolism.
Hydrochlorothiazide
Hydrochlorothiazide is not metabolized but is eliminated rapidly by the kidney through glomerular filtration. At least 61% of the oral dose is eliminated unchanged within 24 hour. The half life of hydrochlorothiazide is approximately 6 hours after administration in combination will candesartan cilexetil, remains unchanged.
Pharmacokinetics in special populations
On administration in elderly patients (older than 60 years) Cmax and AUC of candesartan increased by 55% and 75% respectively. However, the response to blood pressure and occurrence of adverse events are similar in both young and elderly. In patients with mild to moderate renal impairment, Cmax and AUC of candesarta increased by 45 and 65% respectively. Half life of candesartan & hydrochlorothiazide was approximately doubled and prolonged, respectively.
The pharmacokinetics of of the combination has not been investigated in patients less than 18 years of age. It should be used with caution in patients with hepatic impairment.
Pharmacodynamics
Candesartan Cilexetil
Candesartan cilexetil is a prodrug which is rapidly converted to the active drug candesartan. Candesartan is an angiotensin Il receptor antagonist specifically AT1 receptor, with tight binding & slow dissociation from receptor. It has no agonist activity. Candesartan has no effect on ACE or other enzyme system associated with the use of ACE. Candesartan inhibits the pressor effects of angiotensin II infusion in a dose dependent manner. After 1 week of once-daily dosing with 8 mg of candesartan cilexetil, the pressor effect was inhibited by approxomatoly 90% at peak with approximately 50% inhibition persisting for 24 hours. Plasma concentrations of angiotensin l and angiotensin II, and plasma renin activity (PRA), increased in dose dependent manner after single and repeated administration of candesartan cilexetil to a healthy subjects and hypertensive patients.
ACE activity was not altered in healthy subjects after repeated candesartan cilexetil administration. The once-daily administration of up to 16 mg of candesartan cilexetil to healthy subjects did not influence plasma aldosterone concentrations, but a decrease in the plasma concentration of aldosterone was observed when 32 mg of candesartan cilexetil was administered to hypertensive patients. In spite of the effect of candesartan cilexetil on aldosterone secretion, very little effect on serum potassium was observed.
Candesartan does not bind or block other receptors or ion channels, which are important in cardiovascular regulation. In multiple-dose studies with hypertensive patients, there were no clinically significant changes in metabolic function including serum levels of total cholesterol, triglycerides glucose, or uric acid. In a 12-week study of 161 patients with non-insulin-dependent (type 1l) diabetes mellitus and hypertension, there was no change in the level of HbA1c.
Hydrochlorothiazide
Hydrochlorothiazide blocks the reabsorption of sodium, mainly in tubules of kidney and aides the excretion of sodium, chloride & water. During long term therapy, peripheral resistance is reduced which contributes to blood pressure reduction. Hydrochlorothiazide reduces plasma volume and extracellular fluid and decreases cardiac output and blood pressure. After oral administration of Candesartan and Hydrochorothiazide fixed dose combination, diuresis of hydrochlorothiazide component begins within 2 hours, peaks in about 4 hours and lasts about 6 to 12 hours
Uses of Candesartan & Hydrochorothiazide
Candesartan & Hydrochorothiazide is indicated for the treatment of hypertension.
Contraindications of Candesartan & Hydrochorothiazide
The combination is contraindicated in patients who are hypersensitive to any component of this product. Because of the hydrochlorothiazide component, this product is contraindicated in patients with anuria or hypersensitivity to other sulfonamide-derived drugs.
Storage:
Store below 30 °C in a dry place, protect from light.
Chemical Structure of Candesartan
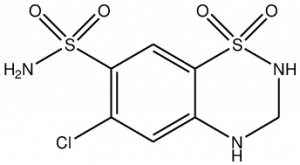
Chemical Structure of Hydrochlorothiazide
2 comments